 The global entropy is not decreased, but information to free energy conversion is possible. 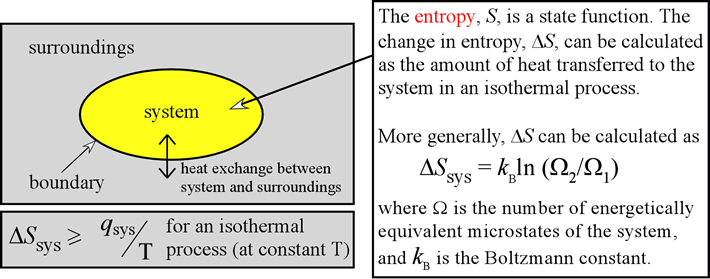
The ratio of the fluctuation entropy and mean Boltzmann, or Gibbs entropy vanishes in the thermodynamic limit for a system of. It is found that their difference is equal to fluctuation entropy, which is a Gibbs-like entropy of macroscopic quantities. Since the Boltzmann entropy can be taken as corresponding to the Clausius entropy if the system is at equilibrium, the former is a generalized form of the. In just the right circumstances therefore, the possession of a single bit of Shannon information (a single bit of negentropy in Brillouin's term) really does correspond to a reduction in the entropy of the physical system. General relationship between mean Boltzmann entropy and Gibbs entropy is established. Of course this does not mean that we understand, whatever is meant by that loaded word 'understand', what time is. The particle can then be left to isothermally expand back to its original equilibrium occupied volume. In this chapter we introduce the statistical definition of entropy as formulated by Boltzmann. 5 Boltzmann's entropy 6 Initial conditions 7 References 8 Recommended reading 9 See also What is time Time is arguably among the most primitive concepts we havethere can be no action or movement, no memory or thought, except in time. S = − k B ∑ i p i ln p i, joules of useful work if the shutter is opened again. This computation shows that Maxwellian distributions are the critical points of the Boltzmann entropy on the affine manifold of densities f corresponding to. Willard Gibbs in the 1870s, is of the form: The defining expression for entropy in the theory of statistical mechanics established by Ludwig Boltzmann and J. Willard Gibbs in the 1870s are similar to the information entropy by Claude Shannon and Ralph Hartley, developed in the 1940s.Įquivalence of form of the defining expressions Boltzmann's grave in the Zentralfriedhof, Vienna, with bust and entropy formula. Boltzmanns macroscopic formulation leads naturally to a formula for the entropy of dilute gases which may be far from LTE. /GettyImages-72308867-1--5814b37b3df78cc2e8229745.jpg)
Later, Gibbs gave another definition of entropy via probabilities of microscopic states of the system. The original concept of entropy was first introduced by the German theoretical physicist Rudolf Clausius into thermodynamics in 1865. 
The mathematical expressions for thermodynamic entropy in the statistical thermodynamics formulation established by Ludwig Boltzmann and J. Boltzmann gave a microscopic definition of entropy, as a logarithm of the number of microscopic states that share the values of physical quantities of the macroscopic state of the system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
